
Проблемы Эволюции |
Science. 2003. V. 302. P.1030-1032.
Increased Longevities of
Post-Paleozoic Marine Genera
After Mass Extinctions
Arnold I. Miller1* and Michael Foote2
I Department of Geology. University of Cincinnati,
Post Office Box 210013, Cincinnati, OH 45221-0013,
USA. 'Department of the Geophysical Sciences. Uni
versity of Chicago, 5734 South Ellis Avenue, Chicago,
IL 60637, USA.
2 To whom correspondence should be addressed E-maiL arnold.millertSuc.edu
Cohorts of marine taxa that originated during recoveries from mass extinctions were commonly more widespread spatially than those originating at other times. Coupted with the recognition of a correlation between the geographic ranges and temporal longevities of marine taxa, this observation predicts that recovery taxa were unusually long-lived geologically. We analyzed this possibility by assessing the longevities of marine genus cohorts that originated in successive substages throughout the Phanerozoic. Results confirm that several mass extinction recovery cohorts were significantly longer lived than other cohorts, but this effect was limited to the post-Paleozoic, suggesting differences in the dynamics of Paleozoic versus post-Paleozoic diversification.
In evaluating the global biotic effects of mass extinctions, attention has focused increasingly on recovery biotas, the taxa that originated or contributed to diversity in the immediate aftermaths of extinction events (/-5). Postextinction diversification has proven to be more geographically and temporally complex than once emisioned, with the onset of major rebounds in diversity characterized by lag times and geographic variability (6-9). Nevertheless, several principles have emerged regarding the biological attributes of recovery taxa. They are thought to be characterized by geographic and/or environmental (i.e., spatial) distributions that were substantially broader, on average, than those of taxa originating at other times {4, 10), although this has not been demonstrated definitively for all of the major mass extinctions of the Phanerozoic. In addition, macroevolutionary theory suggests that taxa with broad spatial distributions should persist for longer geological intervals than those with narrow distributions (//, 12). Thus, it can be hypothesized that cohorts of taxa originating immediately after mass extinctions exhibited significantly greater longevities than their counterparts that originated in surrounding intervals. Here, we demonstrate that this was, indeed, the case for several post-Paleozoic recovery cohorts and that the pattern is impervious to several plausible artifacts. Interestingly, the pattern does not extend back to recovery cohorts of the Paleozoic era.
The analyses were conducted with Sepkoski's genus-level marine compendium {13), a robust resource for ihe investigation of large-scale patterns of diversification and extinction. Sepkoski's "substage"-level stratigraphic binning was maintained for the present study, ;ind only genera whose first and last appearances were resolved to the levels of these subsuge bins were included here {15). The longevity of each genus w;ls calibrated :is the number of bins tor which ii was extant.
Figure 1A depicts the mean longevities of cohorts originating in Phanerozoic intervals ranging from the late Vcndian throuiih the Quaternary. Of particular note are three statistically significant (15) longevity maxima in the post-Paleozoic that correspond to the Early Induan and Late Induan of the Early Tria-ssic: the Late Nonan of the latest Triassic and the adjacent Hettangian of the earliest Jurassic: and the Early Bernasian of the Early Cretaceous {16). In addition, although not as pronounced as the earlier maxima, a local maximum is observed for the Early Paleocene (supporting online text).
A comparison of mean genus longevities with Phanerozoic genus extinction rates (/7) suggests a close relation between longevity maxima and mass extinctions (Fig. IB). Three of the maxima encompass intervals immediately after three of the "big five" mass extinctions of the Phanerozoic: the Late Permian. Late Triassic. and Late Cretaceous (K/T) events. In addition, the Early Bernasian maximum occurs immediately after an interval of elevated extinction (18) in the Late Tithonian.
Before considering the possible biological importance of the observed signal, it is necessary to assess potential nonbiological factors that might have played artifactual roles in producing the outcome. First, a decline analogous to that exhibited in the approach to the Recent might also have been produced in the approach to any major extinction event (supporting online text). To assess this possibility, the analysis was redone after culling all genera that became extinct during the mass extinctions in question, as well as during additional post-Paleozoic extinction events (15).
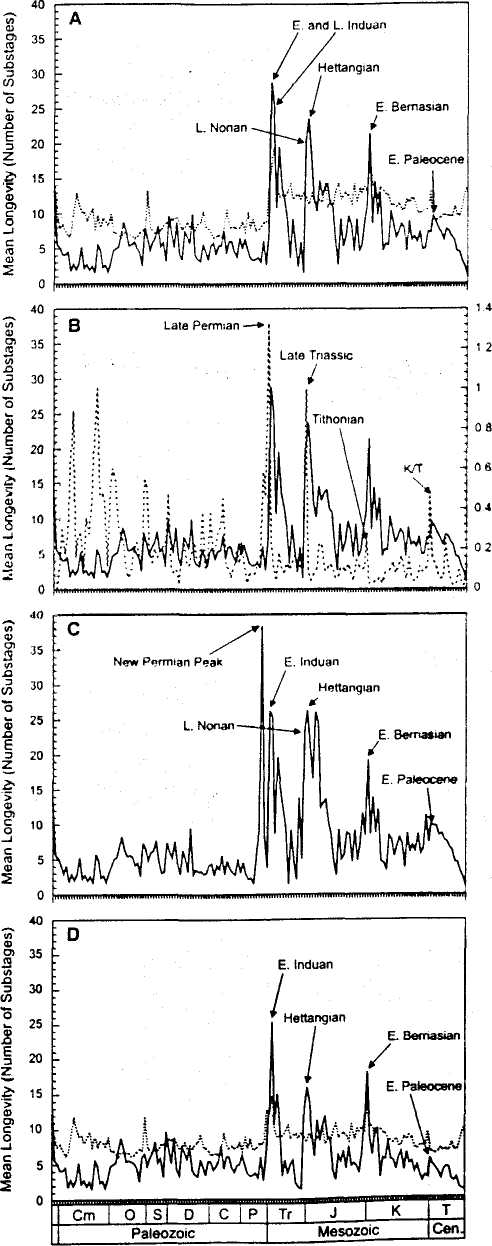
Fig. 1. Mean longevities of genus cohorts originating throughout the Phanerozoic and latest Neoproterozoic (solid lines). (A) Maxima are labeled; statistical significance is indicated when the peak exceeds the upper 95% confidence interval (dotted line) derived with a randomization procedure (75). (B) Mean genus longevities compared with percapita extinction rates (dotted line). Extinction peaks that precede longevity maxima are labeled. (C) The pattern after culling genera that became extinct during several post-Paleozoic mass extinctions (7fi). (D) The pattern after ailing genera that survived to the present day: dotted line as in (A) but based only on genera that did not survive to the present day. Cm, Cambrian: O, Ordovician: S. Silurian: D, Devonian: С Carboniferous: P, Permian: Tr, Triassic J, Jurassic K. Cretaceous; T, Ternary.
The results (Fig. 1С) demonstrate that the initial set of maxima is maintained, although the relative heights of some maxima were altered and. interestingly, an additional maximum is observed for the Late Guadalupian interval of the Late Permian. Second, to investigate the possibility that the means of observed maxima were inflated by the presence of a small number of genera that persisted all the way to the Recent, the analysis was also conducted after culling from the original database all genera that are extant today. The resulting pattern was quite similar to that observed without culling (Fig. ID), although the Hettangian maximum was reduced, and the Early Paleocene maximum is no longer statistically significant.
We have carried out the same analyses using several alternative protocols: the use of median cohort longevities rather than means: the use of stage-level bins rather than substages. which would have permitted die inclusion of genera from the database whose first or last occurrences were not resolved to substage; and the merging together of adjoining substage bins that were either of unusually short duration or contained small cohorts. Although the nature of some maxima changed—particularly the broadening of some postextinetion maxima to include the mass extinctions that immediately preceded them— they did iwt appreciably alter the results reported here, demonstrating that the results are robust.
Figure 1 exhibits two additional features. First, the pattern is almost entirely limited to the post-Paleormc. There is no indication of a meaningful pattern at any time before the Late Guadalupian. nor is there any consistent association between Paleozoic mean longevity patterns and the record of Paleozoic extinction (Fig. ID). Given that the mean duration of Paleozoic sub-stages was greater than that of the post-Paleozoic (15). there might be some concern that the depressed Paleozoic pattern is an artifact: A Paleo-
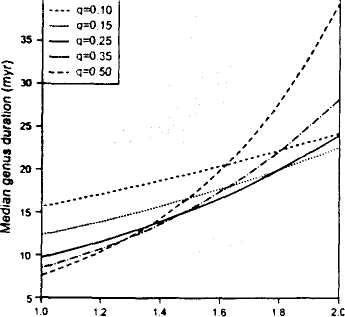
Initial ratio of species origination rate (p) to species extinction rate (q)
Fig. 2. Elevation in median genus duration produced during an interval in which speciation rate (p) within the genus exceeds species extinction rate (q). The initial interval is assumed to be 4 My, about the length of a substage in our analysis.
zoic genus that was extant for the same amount of absolute time as a post-Paleozoic genus might have been extant for a smaller number of sub-stages. However, the actual differences in mean substage lengths between the two intervals [3.72 million years (My) for the Paleozoic versus 3.36 My for the post-Paleozoic] are too small to have caused the longevity differences observed here. Rather, the pattern bolsters the growing evidence suggesting that there were differences in the dynamics of Paleozoic versus post-Paleozoic origination and extinction (19), with the transition perhaps triggered by the Late Permian mass extinction. In addition, preserved biotas came predominantly from epicontinental seas in the Paleozoic but from open ocean environments in the post-Paleozoic (20). This may have caused a secular transition in the evolutionary attributes of marine taxa, given indications that the dynamics of taxa differed noticeably between these two settings (21).
Second, in two cases, the maxima include cohorts that originated in substages during intervals of mass extinction. The peak value observed in the Hettangian was immediately preceded by a value that was nearly as high in the Late Norian. an interval of mass extinction (Fig. ID). In addition, the Late Guadalupian peak that emerged when genera that became extinct during mass extinctions were culled from the data (Fig. 1С) occurred in an interval of elevated extinction that preceded the end-Permian coup de grace (22). Thus, in at least some instances, it appears that genera originating during intervals of elevated extinction that survived these events had longevities, and perhaps other characteristics, comparable to those observed in recovery cohorts. This result is not unexpected, given previous analyses of "disaster taxa" that survived mass extinctions (/-5. /0).
One possible mechanism for the results documented here concerns the effect of species-level origination and extinction rates on the longevities of genera. If. as would be expected, speciation rates are high relative to species extinction rates in the aftermaths of mass extinctions, then a genus that originates shortly after an extinction event, or carries over from before the event, will tend to accumulate species during the substage after the event. All else being equal, this initially higher species richness should enhance the longevity of the genus relative to genera that originate after the interval of elevated speciation rates. To illustrate this mechanism. Fig. 2 shows results from a basic birth-death model (15, 23, 24). Speciation rates within genera are assumed to be higher than species extinction rates for an initial substage; afterward, speciation and extinction rates are assumed equal.
These calculations suggest that a substantial difference in median genus longevity can result from a modest and short-lived excess of speciation over species extinction. For example, the average long-term rate of species extinction of marine invertebrates is in the neighborhood of 0.25 per lineage-million-years (25). In this case, a speciation rate that is 60% higher than extinction for just a few million years, after which it is equal to extinction, will increase median genus duration by —50% relative to a speciation rate that is equal to extinction from the start.
Although our analyses confirm expectations that several recovery cohorts had greater mean longevities than did cohorts originating at other times, they do not demonstrate that these same genera also had unusually large geographic or environmental ranges. This can only be determined by directly evaluating the spatial occurrences of these genera in strata around the world.
Our findings also have ramifications for conservation biology. There is compelling evidence that the earth is currently in the midst of a human-induced mass extinction (26) and that many of the survivors of this extinction are likely to be ecological generalists capable of subsisting in a wide spectrum of settings. The present analysis suggests that the same will be true of any initial recovery of global biodiversity from this extinction and that the taxa that contribute to the recovery are likely therefore to persist for extended periods into the future.
1. J. K. Schubert, D. J. Bottjer, Geology 20, 883 (1992).
2. 0. t RodUnd. 0. ). Bottjer, Paliiot 16. 9S (2001).
3. M. L Droser. 0. J. 8ott|er, P, M. Shethan, C. R.
McChee, Geology 28, 675 (2000),
4. 0. H. Erwin. Proc. NmI. text. Sa U.S.A. 98, 5399 (2001)
5. P. M, Sheehan.,4nn. Rev. fJ/ТЛ H.vwf. Scl. 29. 331 (2001).
6. 0. H. Erwin. Science 279. 1324 (1998).
7. 0, Jablonski. Science 279. 132? (1998).
8. J. W. Kirchner. A Weil Nature 404. 177 (20O0).
9. J. W. Kirchner. Nature 41S. 65 (2002).
10. D. H. Erwin. Trend!. Ecol. 1vol. 13. 344 (1998).
11. D. Jablonski. Science 231. 129 (1986).
12. D. Jablonski. Science 253. 754 (1991).
13. J. J. Sepkoski Jr.. Bull. Am. Paleontol. 363. 1 (2002).
14. J. M. Adrain. 5. R. Westrop, Science 289. 110 (2000).
15. Materials and methods are available as supporting material on Science Online.
16. A similar pattern was derived independently by A. V. Markov (27). but was discussed in a different context from that of the present study.
17. M. Foote. Paleobiology (suppL) 26. 74.
18. D. M. Raup. J. J. Sepkoski Jr., Science 231, 833 (1986).
19. M. Foote. Paleobiology 26, 578 (2000).
20. L J. Walker, B. H. Wilkinson, L С hrany. / Ceoi 110. 75 (2002).
21. P. M. Sheehan. Pafeobio/ogy 1. 205 (1975).
22. S. M. Stanley. X. Yang, Science 266, 1340 (1994).
23. D. С Kendall. Ann. Math. Statist. 19. 1 (1948).
24. О. М. Raup. Paleobiology 11, 42 (1985).
25. D. M. Raup. Paleobiology 17. 37 (1991).
26. N. H. Myers. A. H. Knoll. Proc. Natl. Acad. Sd. U.S.A. 98, 5389 (2001).
27. A. V. Markov, Pateontol. J. 36. 121 (2002).
28 We thank D. M. Raup and D. H. Erwin for discussions, A. Hendy for technical assistance with the figures, and NASA (Program in Exobiology) and NSF (Programs in Biocomplexity and in Geology and Paleontology) for financial support. This is Paleobiology Database Publication No. 20.
Supporting Online Material
wwwjciencemag.org/cgi/content/full/302y5647/1030/
28 July 2003; accepted 22 September 2003